Fungal & Yeast Dermatoses : DERMATOPHYTOSIS
- At a glance
- WHAT DOES IT LOOK LIKE?
- WHAT ELSE LOOKS LIKE THIS?
- HOW DO I DIAGNOSE IT?
- HOW DO I MANAGE IT?
- COMMENTS
AT A GLANCE
- Infection of hair, stratum corneum, and claw caused by protein-consuming fungi of the genuses Microsporum and Trichophyton, most commonly M. canis, M. gypseum, and T. mentagrophytes.
- Furunculosis and subsequent deep dermal and subcutaneous infection is an uncommon presentation.
- M. canis is the most frequent cause of dermatophytosis in the dog and cat. Some individuals, particularly cats, are asymptomatic carriers.
- Dermatophytosis is obtained via contact with infected hair or scale from infected/carrier animals, fomites, or spores in contaminated soils or indoor environments. Infective spores may remain viable in the environment for up to 18 months.
- M. canis is a zoophilic dermatophyte; the primary hosts are cats and dogs. It is of particular concern in shelter environments and multi-pet households.
- M. gypseum is a geophilic dermatophyte, inhabiting soil.
- T. mentagrophytes is transmitted via contact with reservoir hosts (typically rodents) or their environments.
- Incidence of dermatophytosis appears to have geographic variability.
- Immunocompromised, geriatric, and young individuals are at greater risk for infection.
- Genetics may play a role as Persian cats and Yorkshire terriers are over-represented, and some individuals in other breeds appear to be predisposed to recalcitrant or repeat infections.
- Concurrent overgrowth of Staphylococcus species is common.
- Zoonosis is possible with all three species, but most often with M. canis.
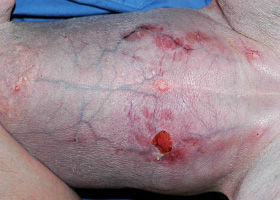
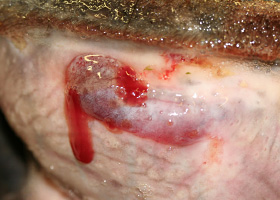
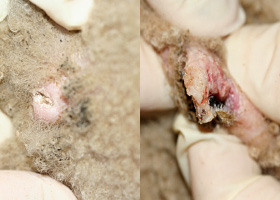
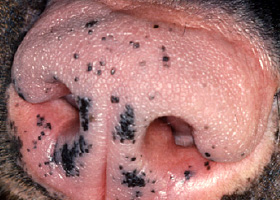
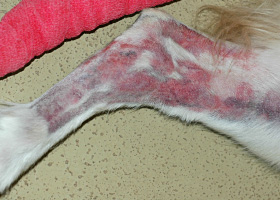
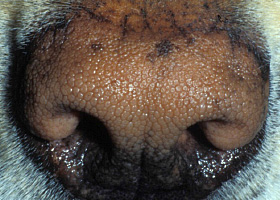
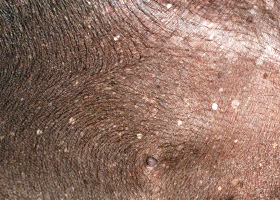
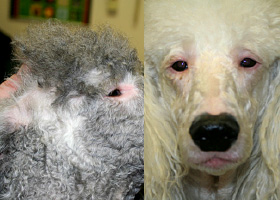
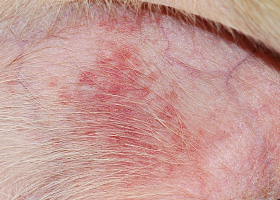
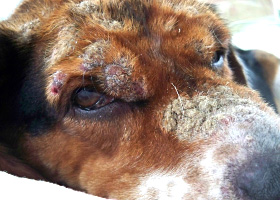
WHAT DOES IT LOOK LIKE?
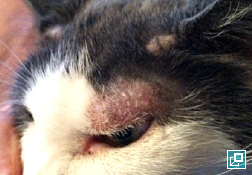
- Feline chin acne
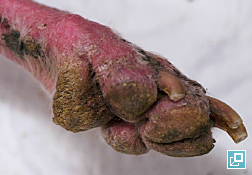
- Nodule
- Draining tracts
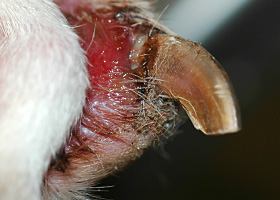
- Onychodystrophy (abnormal shape or texture of the claw)
- Paronychia (claw fold inflammation)
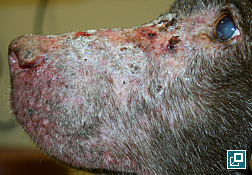
- Presentations may be focal or generalized and may involve the trunk, limbs, tail or facial areas including the pinnae.
- Pruritus is typically mild, but varies from absent to severe.
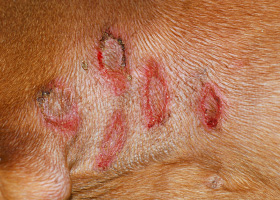
- Lesions may not have a ring-like appearance as is classic in human "ringworm."
- Dermatophytosis is over-diagnosed in dogs, and it is often stated for this species, "If it looks like ringworm, it's probably staphylococcal pyoderma."
WHAT ELSE LOOKS LIKE THIS?
- Staphylococcal pyoderma
- Demodicosis
- Cheyletiellosis
- Sarcoptic mange (pinnal margin and distal limb involvement)
- Feline hypersensitivity syndromes (flea-bite hypersensitivity, cutaneous adverse food reaction, and atopic dermatitis)
- Pemphigus foliaceus
- Vasculitis (involvement of distal extremities)
- Epitheliotropic cutaneous lymphoma
- Nodule: differentials of infectious, neoplastic, sterile inflammatory syndromes, canine acral lick dermatitis, foreign body reaction, and drug/vaccine reaction.
- Onychodystrophy: differentials of bacterial infection, symmetric lupoid onychodystrophy, vasculitis and pemphigus foliaceus.
HOW DO I DIAGNOSE IT?
- Fungal culture is the most sensitive and specific diagnostic technique and is required to determine the species of dermatophyte involved.
- Species identification is recommended to better understand mode of acquisition and to develop strategies for control.
- This technique is insensitive and non-specific for identification of M. canis and is positive in approximately 50% of cases. When positive, it is helpful in selecting areas for culture. However, a negative result does not rule out dermatophyte infection.
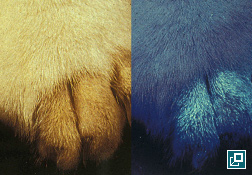
- A Wood's lamp (wavelength 340-450 nm) is used to examine the patient.
- The patient is examined in a dark room with the light several inches from the skin
- Yellow-green fluorescence of the hair is a positive result
- False positive assessments are common as topical products and scale may fluoresce.
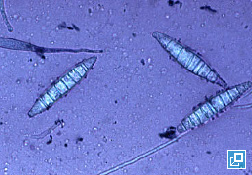
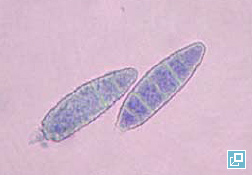
- Hairs are plucked and examined under mineral oil on low power (10X objective).
- Better visualization can be obtained via use of KOH or chlorphenolac as "keratin-clearing agents" instead of mineral oil.
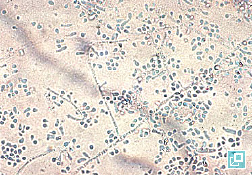
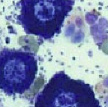
- Presence of hyphae, conidia, or arthrospores is characteristic of dermatophytosis.
- Infected hairshafts may be swollen or frayed with an irregular cortex.
- This technique does not identify the species of dermatophyte.
- This technique is not as sensitive as culture.
- Alcohol is applied to hairs and skin prior to sampling to reduce saprophyte growth.
- Hairs are plucked with forceps from several locations at the periphery of active lesions.
- Hairs are gently applied to dermatophyte test medium, ensuring bulbs are contacting medium.
- The distal ends of long hairs may be trimmed to aid in application prior to, or after plucking.
- Scales and crust are also collected with a clean scalpel blade or flea comb and applied to the medium.
- For suspected onychomycosis, claw shavings may be applied onto the medium.
- This technique is used to screen for carrier status of a patient or to evaluate large areas.
- A sterile toothbrush is used to brush the entire animal for screening, or brushing may be limited to the affected areas
- Bristles are embedded in the medium or scales/hairs teased out onto the medium
- The medium is incubated at room temperature or in an incubator (30°C, 30% humidity).
- The medium is assessed on a daily basis for up to 3 weeks in hospital or at a laboratory.
- Color change of the medium within 24 hours of white/buff colony growth is indicative of a probable dermatophyte.
- Macroconidia are produced several days after colony growth and are used to identify the dermatophyte species.
- To evaluate macroconidia, clear acetate tape may be touched to the surface of the colony, dark stain is applied to a slide and the tape is placed over the stain and examined microscopically on low power (10x objective).
- For nodular presentations, fungal tissue culture may be performed by a diagnostic laboratory, because cultures from hair plucks may be negative.
- Cytology is recommended for nodule and draining tracts as arthrospores may be seen in pyogranulomatous exudates.
- A PCR test for M. canis has been developed with high sensitivity and specificity. It is not commercially available at this time.
- Histopathology may identify dermatophytes, but does not identify the species, thus fungal culture is also recommended.
- Special stains (PAS, GMS) may be used to improve visualization of spores and hyphae.
- Folliculitis and furunculosis with pyogranulomatous dermatitis may be seen, but dermatophytes may not be present in sections examined even with the aid of special stains.
- Histopathologic findings may mimic pemphigus foliaceus and pemphigus erythematosus, including the presence of acantholysis particularly with infection by T. mentagrophytes.
- Histopathology is indicated in nodular lesions. Pyogranulomatous inflammation is often noted, but as with more common presentations, fungal elements may not be present. Deep bacterial and fungal cultures are recommended.
HOW DO I MANAGE IT?
- Treatment involves a multi-pronged approach: topical/and or systemic therapy, environmental management, and in the case of M. canis, assessment for household canine and feline carriers.
- Recheck with repeat culture should be performed 1-3 weeks after initiation of therapy and every 1-3 weeks thereafter. Treatment should be continued until 2-3 negative cultures are obtained.
- Treatment duration is variable and may take from 14 days to 6 months.
- Spontaneous resolution may occur within three months for otherwise healthy patients.
- Topical antifungal therapy is recommended in all cases of dermatophytosis to aid in resolution and to reduce environmental contagion.
- Gentle clipping of the hair will aid in application of topical therapy and remove infected hairshafts. However, clipped hair must be disposed of to prevent environmental contamination.
- Lime sulfur 2-4% dips are an effective, safe, sole therapy for dermatophytosis and are applied once to twice weekly.
- Rinses containing miconazole 2% or enilconazole are effective, and are preferred over sprays and wipes because of better saturation and distribution and should be performed on a once to twice-weekly basis.
- These preparations are best utilized for focal presentations and applied every 12-24 hours.
- Active ingredients include: clotrimazole 1%, miconazole 1-2%, terbinafine 1%, thiabendazole 4%, amphotericin B 3%, enilconazole and nystatin.
- Shampoo therapy has little residual activity and is less effective than other topical therapies.
- Active antifungal ingredients include: ketoconazole 1-2% and miconazole 2%. Preparations may also include chlorhexidine.
- Systemic antifungal treatment is recommended in combination with topical therapy for patients with generalized or multifocal presentations, multi-pet households, long hair coats, nodule, onychomycosis, and focal presentations that fail topical therapy.
- Systemic therapy is selected empirically. Culture and sensitivity of dermatophytes are not routinely performed by laboratories, and reliable, reproducible susceptibility breakpoints are yet to be established and correlated with clinical efficacy.
| MEDICATION | DOSE | NOTES |
|---|---|---|
| Ketoconazole |
|
|
| Itraconazole |
|
|
| Fluconazole |
|
|
| Terbinafine |
|
|
| Griseofulvin |
|
|
- These antifungals may induce hepatotoxicity and should be avoided or used with caution in patients with hepatopathy. Liver enzymes (ALP, ALT) should be assessed every 2-4 weeks.
- Many drug interactions with the azole class are possible and concurrent medications should be evaluated.
- Vomiting and diarrhea may occur with the aforementioned systemic antimicrobials.
- Lufenuron has been previously suggested as a treatment for dermatophytosis, but has not shown to be effective in controlled trials.
ENVIRONMENTAL CONTROL
- M. canis spores may be viable for up to 18 months. Environmental control may reduce reinfection of the patient, humans, and other household animals.
- Lack of control may lead to treatment failure or relapses.
- Consider the Following Measures:
- Culture positive animals should be isolated from negative animals, preferably in an easily-cleaned room devoid of clutter with minimal upholstery
- Treat non-porus surfaces with 1:10 household bleach or enilconazole twice weekly
- Vacuum areas inhabited by positive animals daily, and floors/walls cleaned with electrostatic, microfiber cloths
- Upholstery may be treated with Lysol spray
- Remove/discard bedding and rugs that are difficult to disinfect
- Cleaning of ducts/vents and replacement of furnace filters
- Of note, steam cleaning alone is not effective as temperatures achieved and sustained are not high enough to kill spores.
- Culture of the environment (household or cattery) may be helpful to determine if control measures are effective. Electrostatic cloths or gauze can be wiped on surfaces and then touched to the surface of culture medium three times to inoculate.
- For patients with T. mentagrophytes, reduced exposure to heavily-populated rodent habitats or rodent control is recommended. If rodents are kept as household pets, they may be screened via the MacKensie toothbrush technique
- All household dogs and cats should be screened for carrier status (Mackenzie toothbrush technique), particularly when M. canis has been identified.
- Culture positive animals should be isolated from negative animals. Care and attention should first be provided to non-infected animals to prevent spread.
- Treat exposed and culture-positive animals with lime sulfur 2-4% or systemic antifungals until two negative cultures have been achieved.
- Aforementioned environmental control should be performed.
- Additional information and recommendations for catteries and shelters can be found in the suggested references.
- If T. mentagrophytes is involved, and rodents or rabbits are kept as pets, perform Mackenzie toothbrush technique to screen for carrier status of these pets.
- Instruct clients to wash hands after handling all pets.
- Dermatophyte-positive animals should be isolated from other patients and gloves should be worn during examination. Lab coats should be changed prior to examining other patients.
- Disinfect the examination room with 1:10 bleach or enilconazole.
COMMENTS
- Causes of treatment failure are inadequate treatment duration, failure of environmental control and immunocompromised status, such as FIV or FeLV infection in cats and hyperadrenocorticism in dogs.
- Prognosis is good, with the exception of heavily contaminated environments such as catteries, shelters, and homes with multiple infected animals. Read the suggested references for additional tips on dealing with these situations.
- Instruct clients or staff members in contact with positive patients to consult a physician for diagnosis and treatment should they develop skin lesions.
- Miller, William H., Griffin, Craig E., Campbell, Karen L. Muller & Kirk's Small Animal Dermatology. St. Louis: Elsevier, 2013 243-249.
abscess
A discrete swelling containing purulent material, typically in the subcutis
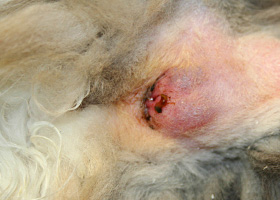
Perianal abscess in a dog
alopecia
Absence of hair from areas where it is normally present; may be due to folliculitis, abnormal follicle cycling, or self-trauma
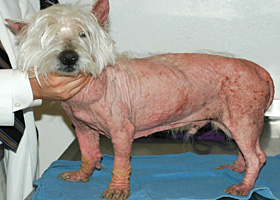
Extensive alopecia secondary to cutaneous epitheliotropic lymphoma
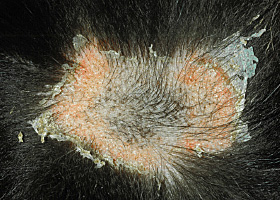
alopecia (“moth-eaten”)
well-circumscribed, circular, patchy to coalescing alopecia, often associated with folliculitis
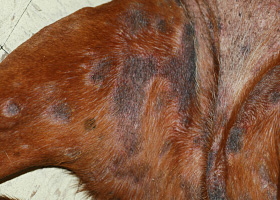
“Moth-eaten” alopecia secondary to superficial bacterial folliculitis
hemorrhagic bullae
Blood-filled elevation of epidermis, >1cm
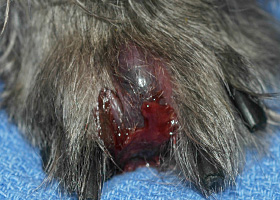
Interdigital hemorrhagic bulla in a dog with deep pyoderma and furunculosis
comedo
dilated hair follicle filled with keratin, sebum
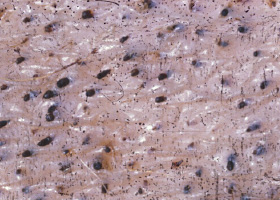
Comedones on the ventral abdomen of a dog with hypercortisolism
crust
Dried exudate and keratinous debris on skin surface
Multifocal crusts due to pemphigus foliaceus
epidermal collarettes
Circular scale or crust with erythema, associated with folliculitis or ruptured pustules or vesicles
Epidermal collarettes in a dog with Staphylococcus superficial bacterial folliculitis
erosion
Defect in epidermis that does not penetrate basement membrane. Histopathology may be needed to differentiate from ulcer.
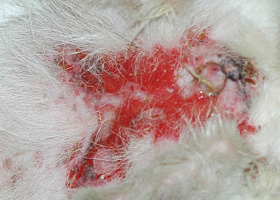
Erosions in a dog with vasculitis
erythema
Red appearance of skin due to inflammation, capillary congestion
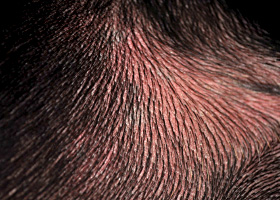
Erythema in a dog with cutaneous drug eruption
eschar
Thick crust often related to necrosis, trauma, or thermal/chemical burn
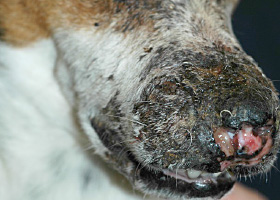
Eschar from physical trauma
excoriation
Erosions and/or ulcerations due to self-trauma
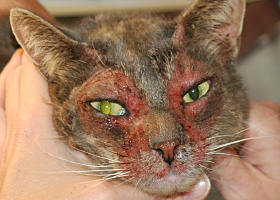
Excoriations in a cat with atopic dermatitis
fissure
Excessive stratum corneum, confirmed via histopathology. This term is often used to describe the nasal planum and footpads.
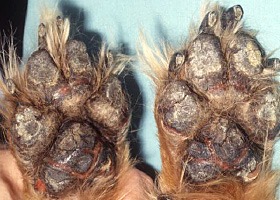
Fissures of the footpads in a dog with superficial necrolytic dermatitis
fistula
Ulcer on skin surface that originates from and is contiguous with tracts extending into deeper, typically subcutaneous tissues
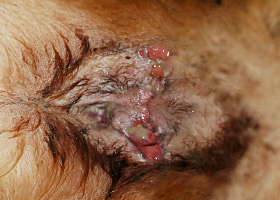
Perianal fistulas in a dog
follicular casts
Accumulation of scale adherent to hair shaft
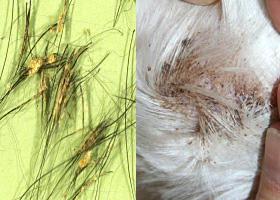
Follicular casts surrounding hairs from a dog with hypothyroidism
hyperkeratosis
Excessive stratum corneum, confirmed via histopathology. This term is often used to describe the nasal planum and footpads.
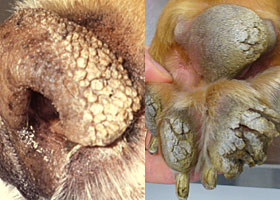
Idiopathic hyperkeratosis of the nasal planum (left) and footpads (right)
hyperpigmentation
Increased melanin in skin, often secondary to inflammation
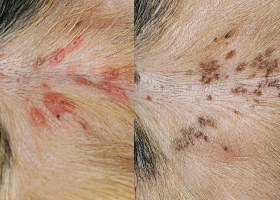
Inflammatory lesions (left) resulting in post-inflammatory hyperpigmentation (right)
hypotrichosis
Lack of hair due to genetic factors or defects in embryogenesis.

Congenital hypotrichosis in chocolate Labrador puppies.
lichenification
Thickening of the epidermis, often due to chronic inflammation resulting in exaggerated texture
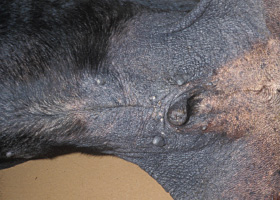
Lichenification of skin in a dog with chronic atopic dermatitis and Malassezia dermatitis
macule
Flat lesion associated with color change <1cm
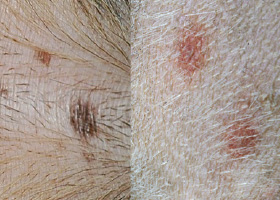
Pigmented macule (left) Erythematous macule (right)
melanosis
Increased melanin in skin, may be secondary to inflammation.
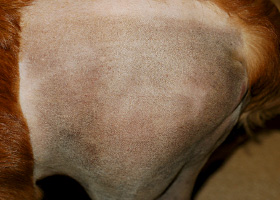
Post inflammatory hyperpigmentation of this dog’s thigh
miliary
Multifocal, papular, crusting dermatitis; a descriptive term, not a diagnosis
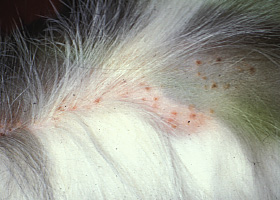
Miliary dermatitis in a flea allergic cat
morbiliform
A erythematous, macular, papular rash; the erythematous macules are typically 2-10 mm in diameter with coalescence to form larger lesions in some areas
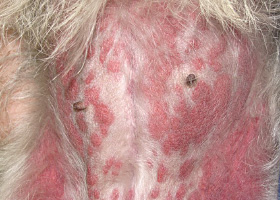
Morbiliform eruptions in a dog with a cutaneous drug reaction
onychodystrophy
Abnormal nail morphology due to nail bed infection, inflammation, or trauma; may include: Onychogryphosis, Onychomadesis, Onychorrhexis, Onychoschizia
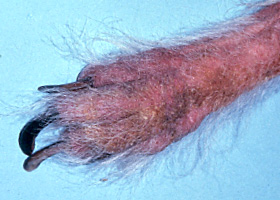
Onychodystrophy in dog with chronic allergies
onychogryphosis
Abnormal claw curvature; secondary to nail bed inflammation or trauma

Onychogryphosis in a dog with symmetric lupoid onychodystrophy
onychomadesis
Claw sloughing due to nail bed inflammation or trauma
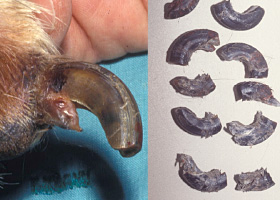
Onychomadesis in a dog with symmetric lupoid onychodystrophy
onychorrhexis
Claw fragmentation due to nail bed inflammation or trauma
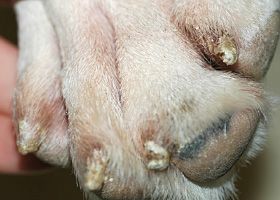
Onychorrhexis in a dog with symmetric lupoid onychodystrophy
onychoschizia
Claw splitting due to nail bed inflammation or trauma
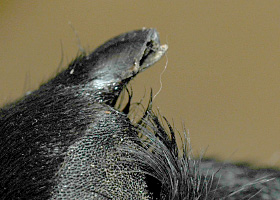
Onychoschizia in a dog with symmetric lupoid onychodystrophy
patch
Flat lesion associated with color change >1cm
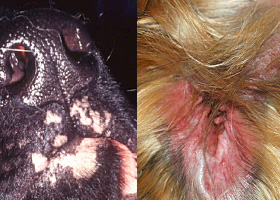
Hypopigmented patch (left), erythematous patch (right)
petechiae
Small erythematous or violaceous lesions due to dermal bleeding
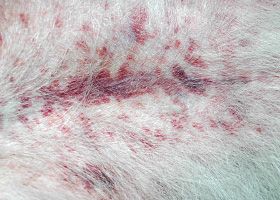
Petechiae in a dog with cutaneous vasculitis
phlebectasia
Venous dilation; most commonly associated with hypercortisolism
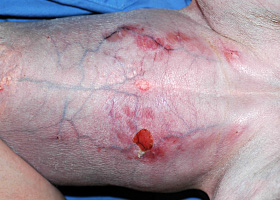
Phlebectasia and cutaneous atrophy due to hypercortisolism in a dog
plaques
Flat-topped elevation >1cm formed of coalescing papules or dermal infiltration
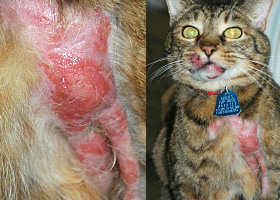
Plaques in a cat with cutaneous lymphoma
pustule
Raised epidermal infiltration of pus
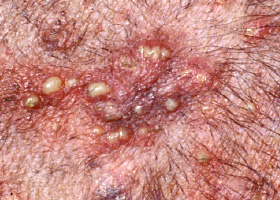
Pustules on the abdomen of a dog with superficial staphylococcal pyoderma.
scale
Accumulation of loose fragments of stratum corneum
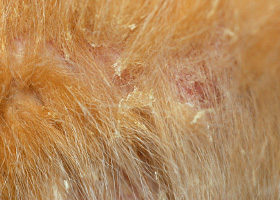
Loose, large scales due to ichthyosis in a Golden Retriever
scar
Fibrous tissue replacing damaged cutaneous and/or subcutaneous tissues
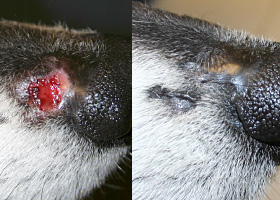
Scarring (right) following the healing of an ulcer (left) in a dog with sterile nodular dermatitis
serpiginous
Undulating, serpentine (snake-like) arrangement of lesions

Serpiginous urticarial lesions on a horse
telangiectasia
Permanent enlargement of vessels resulting in a red or violet lesion (rare)
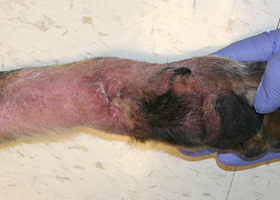
Telangiectasia in a dog with angiomatosis
ulcer
A defect in epidermis that penetrates the basement membrane. Histopathology may be needed to differentiate from an erosion.
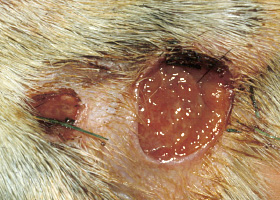
Ulcerations of the skin of a dog with vasculitis.
urticaria
Wheals (steep-walled, circumscribed elevation in the skin due to edema ) due to hypersensitivity reaction

Urticaria in a horse
vesicle
Fluid-filled elevation of epidermis, <1cm
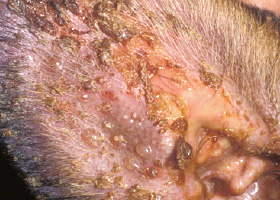
Vesicles and bullae on ear pinna due to bullous pemphigoid
wheal
Steep-walled, circumscribed elevation in the skin due to edema

Wheals associated with intradermal allergy testing in a horse