NEOPLASTIC DERMATOSES : MAST CELL TUMORS – CANINE
- At a glance
- WHAT DOES IT LOOK LIKE?
- WHAT ELSE LOOKS LIKE THIS?
- HOW DO I DIAGNOSE IT?
- HOW DO I MANAGE IT?
- COMMENTS
AT A GLANCE
- Mast cell tumors (MCTs) are neoplasms that that typically develop in the skin and subcutaneous tissues, usually on the trunk and limbs of dogs; they represent the second most common tumor found in dogs.
- Breeds of dog predisposed to develop MCT include those of bulldog descent (Boxers, Pugs, etc), Labrador and Golden Retrievers, and Shar-peis.
- MCT exhibit a wide range in biologic behavior from benign disease, that is readily cured with surgical removal, to aggressive tumors that metastasize widely resulting in death of affected dogs.
- While the molecular changes that drive MCT in many dogs are not known, 25-50% of aggressive MCT possess a mutation in a cell surface receptor called KIT that drives the uncontrolled growth of the malignant cells in these tumors.
- There are now several new therapies available to treat aggressive MCT in dogs and long-term outcome has been significantly improved by using a multimodal approach to treatment including surgery, radiation therapy, chemotherapy and small molecule inhibitors that target KIT.
WHAT DOES IT LOOK LIKE?
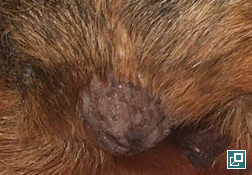
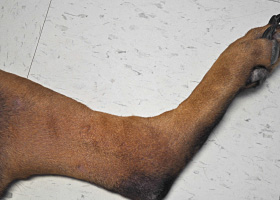
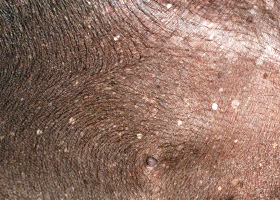
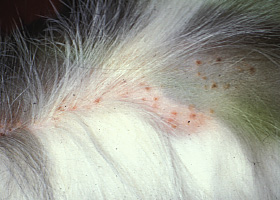
- MCT most commonly appear as a solitary, haired to hairless cutaneous nodule ranging from 1-3 cm in size (Pathologic Image Library - Figure 1).
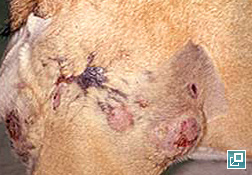
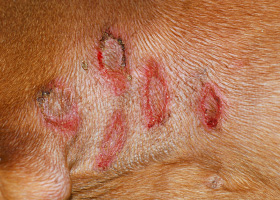
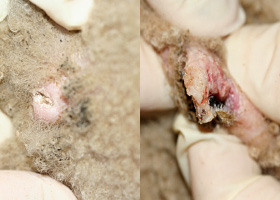
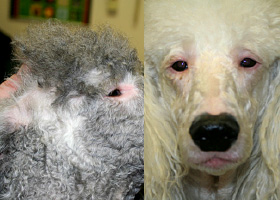
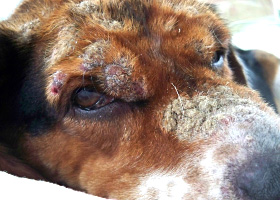
- Some dogs will present with multiple (2-3) MCTs although this does not necessarily affect the overall prognosis (Pathologic Image Library - Figure 2).
- Occasionally owners will report that the tumor appears to increase then decrease in size over several days; this change appearance is often secondary to histamine release from the malignant mast cells causing local swelling.
- As the tumors can be pruritic, trauma from scratching or licking can cause ulcer and less commonly secondary infection.
- Mast cells contain a number of mediators including cytokines, chemokines, prostaglandins, proteases as well as histamine and heparin that they may release. Increased circulating levels in histamine can predispose to the development of gastric ulcer, and affected dogs may present with a history of vomiting and/or diarrhea.
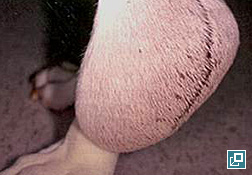
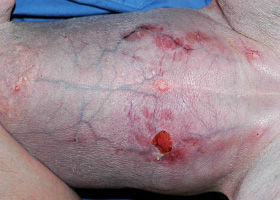
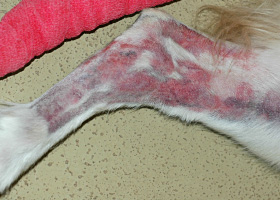
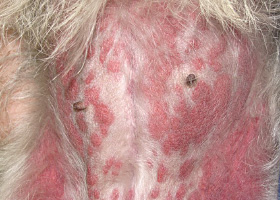
- MCTs may remain unchanged in size over many months to years in dogs; typically this indicates a more benign behavior; in contrast, those tumors that grow rapidly tend to exhibit aggressive biologic behavior and result in metastatic disease (Pathologic Image Library - Figure 3).
WHAT ELSE LOOKS LIKE THIS?
- Fibropapilloma/sebaceous adenoma
- Epidermal inclusion cyst
- Non-healing wound
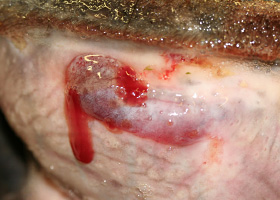
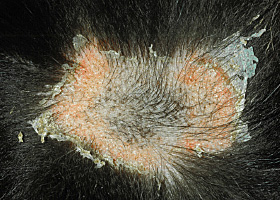
- Lipoma (Pathologic Image Library - Figure 4)
HOW DO I DIAGNOSE IT?
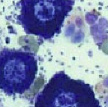
- Cytologic evaluation of fine needle aspirates is the easiest method to diagnose a MCT; poorly differentiated MCTs may contain few, if any granules, necessitating special stains. Additionally, sometimes mast cell granules do not stain with Diff-Quick, making the use of a Wright-Giemsa stain necessary for definitive diagnosis.
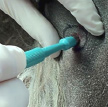
- If cytologic diagnosis proves difficult, a needle or punch biopsy can be obtained; this is preferred to a larger incisional biopsy as release of mast cell mediators can result in impaired clotting and delayed wound healing.
- Excisional biopsy is required for histologic grading which is based on several features including degree of invasion, mitotic index, cellular atypical, etc.
- Staging of dogs with MCT should include standard tests such as CBC, biochemistry profile, and urinalysis. Additionally, the regional lymph nodes should be evaluated by fine needle aspiration (prior to tumor removal) for evidence of metastasis. Lastly, abdominal ultrasound and thoracic radiographs should be performed in dog whose MCTs exhibit signs of biologically aggressive behavior.
- It is important to note that fine needle aspirates of the liver and spleen should be obtained in all dogs with aggressive MCT as these organs may have a normal ultrasonographic appearance despite the presence of metastatic disease.
HOW DO I MANAGE IT?
- There are several prognostic indicators that help to guide the type of therapy for a particular MCT. These include:
- Breed: Pugs and Boxers tend to develop benign MCT while Shar-peis tend to develop aggressive MCT
- Clinical signs: dogs with vomiting, diarrhea, weakness tend to have a worse prognosis
- Growth rate: MCT unchanged in appearance for > 6 months are generally less aggressive
- Stage: dogs with multiple independent MCT may not have worse prognosis, but significant lymph node involvement (i.e., enlarged, effaced nodes) and/or abdominal organ involvement indicates aggressive disease.
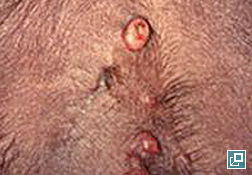
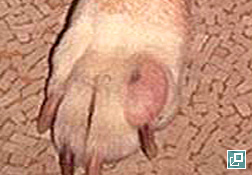
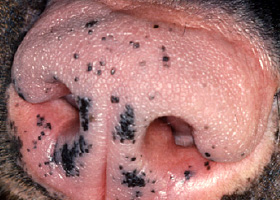
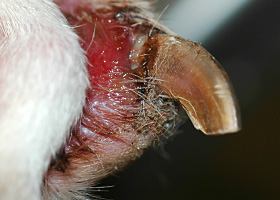
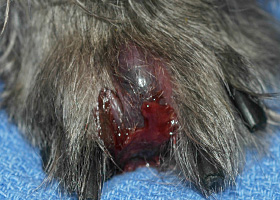
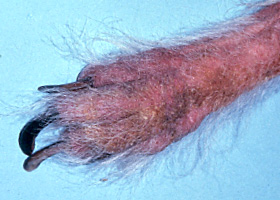
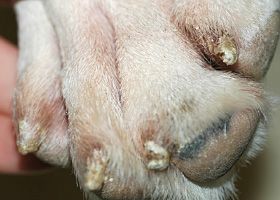
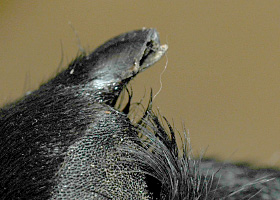
- Location: MCT in the subcutaneous tissues are often less aggressive; those on the muzzle and nail bed (Pathologic Image Library - Figure 5) have a high metastatic rate.
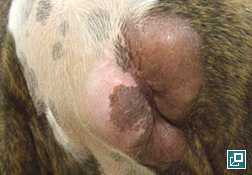
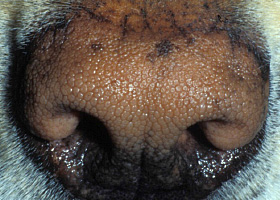
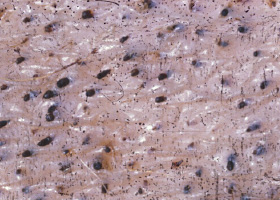
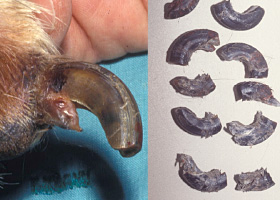
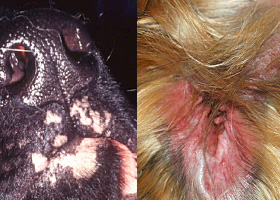
- Grade: grade 1 tumors are typically benign, while grade 3 tumors are typically malignant; about 75% of grade 2 tumors (Pathologic Image Library - Figure 6) will behave aggressively. A new tumor grading system (awaiting validation) has been developed to guide decision making, this system separates MCT into low or high grade tumors.
- Proliferation markers: mitotic index, Ki67 and others are often helpful in determining the likelihood for a MCT to spread.
- KIT mutation: testing is now offered through multiple diagnostic labs; the mutation is associated with a higher risk of local recurrence, metastasis and death.
- The mainstay of MCT treatment is wide surgical excision (2-3 cm lateral margins, 1 fascial plane deep).
- Radiation therapy (RT) is extremely effective at eliminating residual tumor cells following an incomplete excision; recent evidence suggests that RT may also be effective in controlling MCT that are not amenable to surgical removal.
- The use of chemotherapy is indicated following removal of grade 3 MCTs, metastatic MCTs, non-resectable MCT, or for any other MCT with negative prognostic indicators.
- Prednisone: this drug has activity against most MCT, with up to 75% decreasing in size following treatment. It is generally included in all treatment protocols as it also helps to decrease mast cell mediator release.
- Vinblastine: this is the most commonly used chemotherapy agent for MCTs.
- Alkylating agents such as lomustine, chlorambucil and hydroxurea also have activity against MCTs, although these are generally considered to be less effective than that of vinblastine.
- Small molecule inhibitors of KIT, such as toceranib phosphate (Palladia) are indicated for the treatment of recurrent MCT with KIT mutations (70-90% response rate); toceranib may also have activity against MCT without KIT mutation (30% response rate).
- Supportive care including a proton pump inhibitor to decrease gastric acid production and an H1 antagonist to block histamine effects on smooth muscle is indicated in all dogs with aggressive MCT. Sucralfate can also be used if GI ulcer is suspected.
COMMENTS
- The integration of multiple therapeutic approaches (surgery, RT, chemo, and KIT inhibitors) to treat aggressive MCT has substantially improved survival times of affected dogs.
- Given the high response rate of MCTs with KIT mutation to KIT inhibitors such as toceranib, KIT mutation testing should be performed in all dogs with aggressive disease or negative prognostic indicators.
- Blackwood L, Murphy S, Buracco P, De Vos JP, De Fornel-Thibaud P, Hirschberger J, Kessler M, Pastor J, Ponce F, Savary-Bataille K, Argyle DJ. European consensus document on mast cell tumours in dogs and cats. Vet Comp Oncol. 2012 Sep;10(3):e1-e29.
- Kiupel M, Webster JD, Bailey KL, et al. Proposal of a 2-tier histologic grading system for canine cutaneous mast cell tumors to more accurately predict biological behavior. Vet Pathol 2011;48:147-155.
- London CA. Tyrosine kinase inhibitors in veterinary medicine. Top Companion Anim Med 2009;24:106-112.
- London CA, Seguin B. Mast cell tumors in the dog. Vet Clin North Am Small Anim Pract 2003;33:473-489.
- Thamm DH, Turek MM, Vail DM. Outcome and prognostic factors following adjuvant prednisone/vinblastine chemotherapy for high-risk canine mast cell tumour: 61 cases. J Vet Med Sci 2006;68:581-587.
- Mayer MN. Radiation therapy for canine mast cell tumors. Can Vet J. 2006 Mar;47(3):263-5
abscess
A discrete swelling containing purulent material, typically in the subcutis
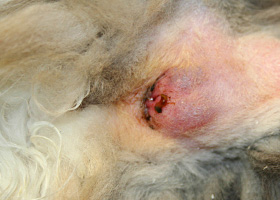
Perianal abscess in a dog
alopecia
Absence of hair from areas where it is normally present; may be due to folliculitis, abnormal follicle cycling, or self-trauma
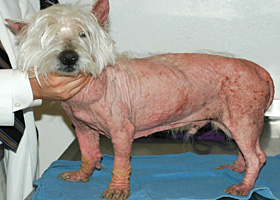
Extensive alopecia secondary to cutaneous epitheliotropic lymphoma
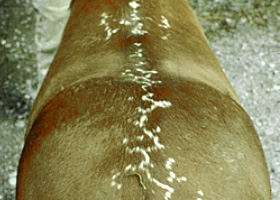
alopecia (“moth-eaten”)
well-circumscribed, circular, patchy to coalescing alopecia, often associated with folliculitis
“Moth-eaten” alopecia secondary to superficial bacterial folliculitis
hemorrhagic bullae
Blood-filled elevation of epidermis, >1cm
Interdigital hemorrhagic bulla in a dog with deep pyoderma and furunculosis
comedo
dilated hair follicle filled with keratin, sebum
Comedones on the ventral abdomen of a dog with hypercortisolism
crust
Dried exudate and keratinous debris on skin surface
Multifocal crusts due to pemphigus foliaceus
epidermal collarettes
Circular scale or crust with erythema, associated with folliculitis or ruptured pustules or vesicles
Epidermal collarettes in a dog with Staphylococcus superficial bacterial folliculitis
erosion
Defect in epidermis that does not penetrate basement membrane. Histopathology may be needed to differentiate from ulcer.
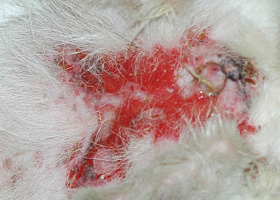
Erosions in a dog with vasculitis
erythema
Red appearance of skin due to inflammation, capillary congestion
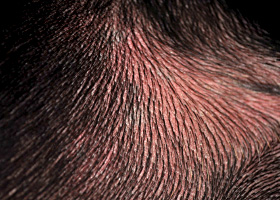
Erythema in a dog with cutaneous drug eruption
eschar
Thick crust often related to necrosis, trauma, or thermal/chemical burn
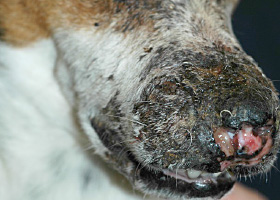
Eschar from physical trauma
excoriation
Erosions and/or ulcerations due to self-trauma
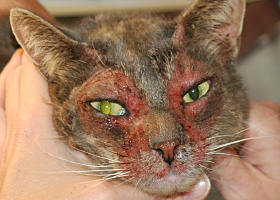
Excoriations in a cat with atopic dermatitis
fissure
Excessive stratum corneum, confirmed via histopathology. This term is often used to describe the nasal planum and footpads.
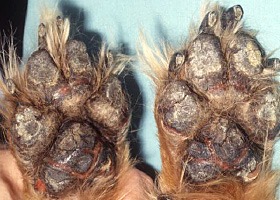
Fissures of the footpads in a dog with superficial necrolytic dermatitis
fistula
Ulcer on skin surface that originates from and is contiguous with tracts extending into deeper, typically subcutaneous tissues
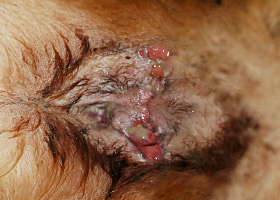
Perianal fistulas in a dog
follicular casts
Accumulation of scale adherent to hair shaft
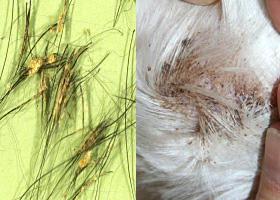
Follicular casts surrounding hairs from a dog with hypothyroidism
hyperkeratosis
Excessive stratum corneum, confirmed via histopathology. This term is often used to describe the nasal planum and footpads.
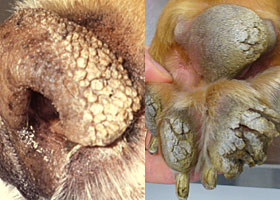
Idiopathic hyperkeratosis of the nasal planum (left) and footpads (right)
hyperpigmentation
Increased melanin in skin, often secondary to inflammation
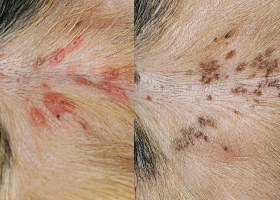
Inflammatory lesions (left) resulting in post-inflammatory hyperpigmentation (right)
hypotrichosis
Lack of hair due to genetic factors or defects in embryogenesis.

Congenital hypotrichosis in chocolate Labrador puppies.
lichenification
Thickening of the epidermis, often due to chronic inflammation resulting in exaggerated texture
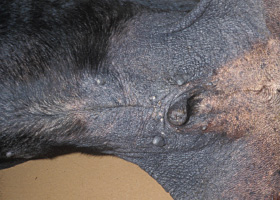
Lichenification of skin in a dog with chronic atopic dermatitis and Malassezia dermatitis
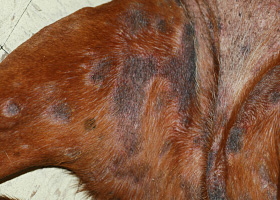
macule
Flat lesion associated with color change <1cm
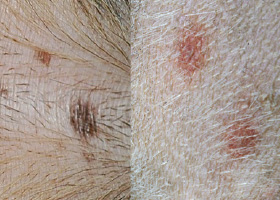
Pigmented macule (left) Erythematous macule (right)
melanosis
Increased melanin in skin, may be secondary to inflammation.
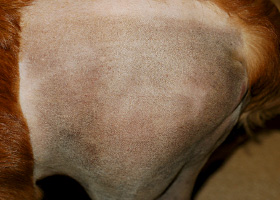
Post inflammatory hyperpigmentation of this dog’s thigh
miliary
Multifocal, papular, crusting dermatitis; a descriptive term, not a diagnosis
Miliary dermatitis in a flea allergic cat
morbiliform
A erythematous, macular, papular rash; the erythematous macules are typically 2-10 mm in diameter with coalescence to form larger lesions in some areas
Morbiliform eruptions in a dog with a cutaneous drug reaction
onychodystrophy
Abnormal nail morphology due to nail bed infection, inflammation, or trauma; may include: Onychogryphosis, Onychomadesis, Onychorrhexis, Onychoschizia
Onychodystrophy in dog with chronic allergies
onychogryphosis
Abnormal claw curvature; secondary to nail bed inflammation or trauma

Onychogryphosis in a dog with symmetric lupoid onychodystrophy
onychomadesis
Claw sloughing due to nail bed inflammation or trauma
Onychomadesis in a dog with symmetric lupoid onychodystrophy
onychorrhexis
Claw fragmentation due to nail bed inflammation or trauma
Onychorrhexis in a dog with symmetric lupoid onychodystrophy
onychoschizia
Claw splitting due to nail bed inflammation or trauma
Onychoschizia in a dog with symmetric lupoid onychodystrophy
patch
Flat lesion associated with color change >1cm
Hypopigmented patch (left), erythematous patch (right)
petechiae
Small erythematous or violaceous lesions due to dermal bleeding
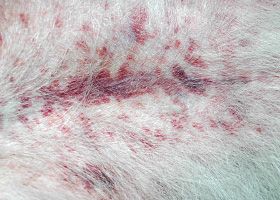
Petechiae in a dog with cutaneous vasculitis
phlebectasia
Venous dilation; most commonly associated with hypercortisolism
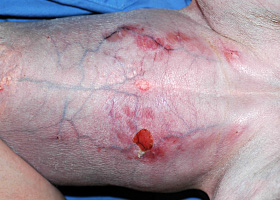
Phlebectasia and cutaneous atrophy due to hypercortisolism in a dog
plaques
Flat-topped elevation >1cm formed of coalescing papules or dermal infiltration
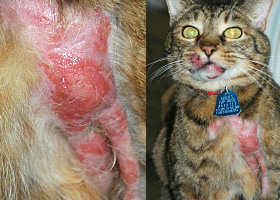
Plaques in a cat with cutaneous lymphoma
pustule
Raised epidermal infiltration of pus
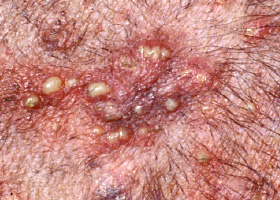
Pustules on the abdomen of a dog with superficial staphylococcal pyoderma.
scale
Accumulation of loose fragments of stratum corneum
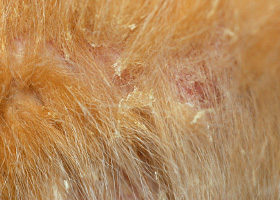
Loose, large scales due to ichthyosis in a Golden Retriever
scar
Fibrous tissue replacing damaged cutaneous and/or subcutaneous tissues
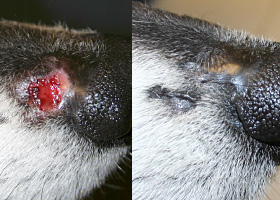
Scarring (right) following the healing of an ulcer (left) in a dog with sterile nodular dermatitis
serpiginous
Undulating, serpentine (snake-like) arrangement of lesions

Serpiginous urticarial lesions on a horse
telangiectasia
Permanent enlargement of vessels resulting in a red or violet lesion (rare)
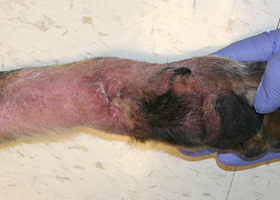
Telangiectasia in a dog with angiomatosis
ulcer
A defect in epidermis that penetrates the basement membrane. Histopathology may be needed to differentiate from an erosion.
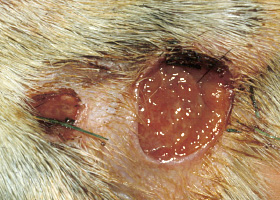
Ulcerations of the skin of a dog with vasculitis.
urticaria
Wheals (steep-walled, circumscribed elevation in the skin due to edema ) due to hypersensitivity reaction

Urticaria in a horse
vesicle
Fluid-filled elevation of epidermis, <1cm
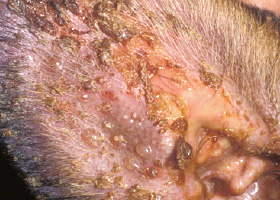
Vesicles and bullae on ear pinna due to bullous pemphigoid
wheal
Steep-walled, circumscribed elevation in the skin due to edema

Wheals associated with intradermal allergy testing in a horse