ENDOCRINE & METABOLIC DERMATOSES : HYPERADRENOCORTICISM - CANINE
- At a glance
- WHAT DOES IT LOOK LIKE?
- WHAT ELSE LOOKS LIKE THIS?
- HOW DO I DIAGNOSE IT?
- HOW DO I MANAGE IT?
- COMMENTS
AT A GLANCE
- Hyperadrenocorticism (HAC) in the dog is a spontaneous disease arising most commonly from a corticotropin secreting, pituitary adenoma that results in bilateral adrenal gland hyperplasia and excessive production of cortisol [pituitary dependent form of HAC (PDH)]. Hyperadrenocorticism can also occur as a result of a cortisol secreting adrenal tumor or adrenal dependent HAC.
- Hypercortisolemia also develops when dogs are administered excessive exogenous sources of glucocorticoids in the management of immune mediated disease or hypersensitivities. Iatrogenic hypercortisolemia is not synonymous with HAC. However all causes of hypercortisolemia can be correctly referred to as Cushing's syndrome.
- The skin is particularly sensitive to increases in cortisol and a number of characteristic changes can occur because of both the protein catabolic and antimitotic effects of glucocorticoids. Glucocorticoids cause cornification abnormalities, inhibit both fibroblast proliferation and collagen production, and cause pilosebaceous gland atrophy. These changes can account for many of the cutaneous clinical signs seen in dogs with HAC.
What does it look like?
- Middle aged to older dogs (median age at time of diagnosis is 10 yrs). Dogs with adrenal dependent HAC are typically older than those with PDH; 75% of PDH and 90% of dogs with adrenocortical tumors are > 9 years of age at time of diagnosis.
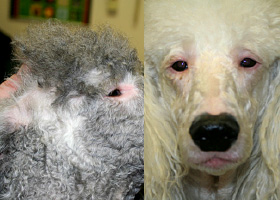
- Typically HAC is diagnosed in small breed dogs and 75% are less than 20 Kg in body weight. However, 50% of dogs with adrenal dependent HAC are greater than 20 kg. Poodles, Dachsunds, Boxers and a number of terrier breeds are predisposed to develop PDH.
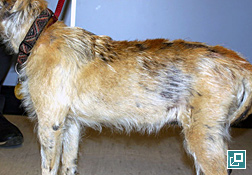
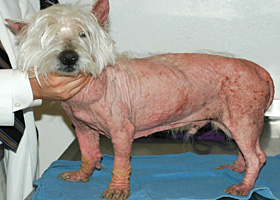
- The most common cutaneous clinical sign seen is alopecia. Non-cutaneous clinical signs are typically also present in dogs with HAC. Commonly seen clinical signs include polyuria and polydipsia, polyphagia, a pendulous abdomen due to muscle wasting, hepatomegaly, muscle weakness and/or atrophy, reproductive changes (anestrus in intact female dogs, clitoral hypertrophy and testicular atrophy), panting, hypertension and peripheral neuropathies (facial paralysis). In some cases, skin changes may be the first presenting clinical sign leading to the diagnosis of HAC without other more typical clinical signs being present.
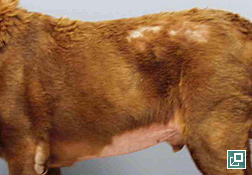
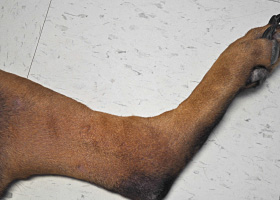
- Alopecia often begins over pressure points and most often involves the trunk in a bilaterally symmetric distribution, but it can present as generalized hair thinning or patchy truncal alopecia.
- The persisting hair coat is often dry, brittle and can be either dull or faded in color reflecting persistence of hair follicles in telogen and a failure to have anagen initiated. This can also explain failure of clipped hair to regrow as quickly.
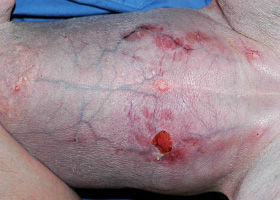
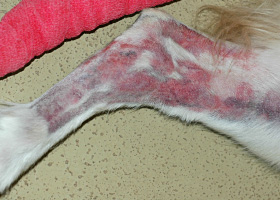
- Hypercortisolemia (endogenous or exogenous) results in thin, hypotonic skin that is easily bruised. This thin hypotonic skin is most evident on the ventral abdomen where atrophy of dermal collagen makes dermal vasculature easily visible.
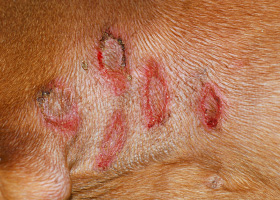
- Hyperpigmentation, cornification disturbances (seborrhea), phlebectasias (dilations or varicosities of the small venules in the superficial dermis), comedones (plug of keratin and sebum within a hair follicle that is blackened at the surface), milia (white, keratin filled with no opening to the skin surface) and striae (irregular areas of skin that look like bands or lines) are all skin changes that can occur as a result of hypercortisolemia.
- Delayed wound healing and increased risk for infections are also consequences of hypercortisolemic changes to the skin
- Bacterial and fungal skin infections can develop in dogs with hypercortisolemia.
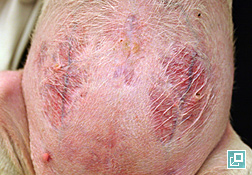
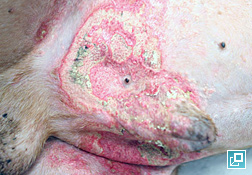
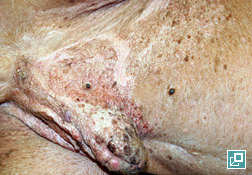
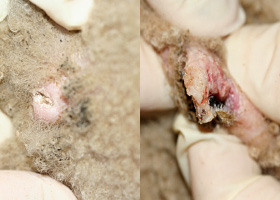
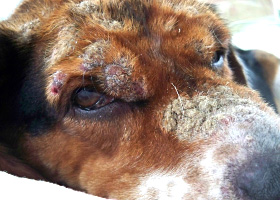
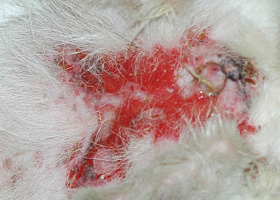
- Calcinosis cutis is a dystrophic calcification seen in dogs with endogenous or exogenous hypercortisolemia. Labrador Retrievers, Rottweilers, Boxers and Pit Bull Terriers were overrepresented breeds in one study. That same study showed the mineral present within the calcinosis cutis lesions to be apatite crystals. Lesions of calcinosis cutis often progress to coalesce into firm, gritty plaques that may ulcerate and develop hemorrhagic crust. Clinically these lesions most often develop over the dorsum or in the inguinal area. Dystrophic calcification can also involve mucosal membranes and the tongue.
What else looks like it?
- Iatrogenic hypercortisolemia from exogenous parenteral and/or topical corticosteroid administration or application.
- Alopecia secondary to canine hypothyroidism
- Alopecia and hyperpigmentation secondary to sex hormone imbalances from testicular or adrenal neoplasia
- Hair cycle arrest in plush coated breeds (alopecia X, adrenal-like hyperplasia)
How do I diagnose it?
- Hyperadrenocorticism is diagnosed based on screening tests performed on dogs with compatible history and physical exam findings along with an awareness of any history of exogenous administration of corticosteroids and concurrent illnesses.
- If a complete blood cell count and a serum biochemistry panel are performed the index of clinical suspicion for HAC is increased if elevated liver enzymes are documented (particularly alkaline phosphatase which has a steroid induced isoenzyme). Other clinicopathologic abnormalities that may be seen in dogs with hypercortisolemia include leukocytosis with a mature neutrophilia, lymphopenia and eosinopenia; thrombocytosis; erythrocytosis; hypercholesterolemia; hypertriglyceridemia; hyperphosphatemia and a decreased blood urea niturogen (BUN). Diabetes mellitus may occur concurrently with HAC and if concurrently present it is typically difficult to control.
- Urinalysis often documents a urine specific gravity that is below 1.020. Proteinuria with a mildly elevated urine protein to creatinine ratio may be present. Urinary tract infections are common and a urine sediment exam may document bacteruria without concurrent pyuria.
- Diagnostic imaging may include radiography, abdominal ultrasound or advanced imaging such as computed tomography (CT) or magnetic resonance imaging (MRI). Abdominal radiographic changes include hepatomegaly and possibly calcification of an adrenal mass (about 50% of adrenal adenomas or carcinomas have some degree of calcification). Abdominal ultrasound may reveal bilateral or unilateral adrenomegaly. Rarely bilateral adrenomegaly is documented with a concurrent adrenal mass. Presence of bilateral adrenomegaly without an adrenal mass can differentiate PDH from an adrenal tumor causing HAC. Computed tomography or MRI are both used to image adrenal glands. MRI is also used in evaluation for possible macroadenomas of the pituitary gland.
- Endocrine function testing to evaluate for HAC involves using one or more screening tests and if positive then differentiating the cause of HAC. Screening tests include a urinary cortisol to creatinine ratio (UCCR), an ACTH stimulation test or a low dose dexamethasone suppression test (LDDST).
- The sample for a UCCR should be obtained in the morning preferably in the dog’s home environment. The test is highly sensitive but has low specificity as numerous other diseases can increase the UCCR. The UCCR is most valuable is in ruling out the diagnosis of HAC because a normal result is not compatible with a diagnosis of HAC.
- An ACTH stimulation test measures the adrenal glands response to a maximal dose of ACTH. There are several different protocols depending on the form of ACTH used and readers are referred to veterinary internal medicine textbooks. Compared to the LDDST the ACTH stimulation test has a couple of disadvantages: it is less sensitive, more expensive and it cannot differentiate PDH from adrenal tumor causing HAC. It can however differentiate iatrogenic hypercortisolemia from HAC, may allow evaluation of other adrenal hormones in patients where disturbances in other adrenal steroid hormones are suspected and it is a shorter test to perform than the LDDST.
- The LDDST is typically considered the preferred screening test to diagnose HAC in the dog as it much more sensitive than the ACTH stimulation test but it cannot identify cases of iatrogenic hypercortisolemia. The specificity of this test is however low and nonadrenal illness may alter the LDDST results. If a dog has cortisol suppression at 4hrs on a LDDST which then normalizes at 8 hrs this is diagnostic for PDH and further differentiating testing is not required. However not all dogs with PDH have suppressed cortisol at 4 hrs and in those cases where there is partial to no suppression additional diagnostic tests are needed to differentiate PDH from an adrenal tumor causing HAC.
- HAC caused by PDH vs. an adrenal tumor may be differentiated based on the LDDST results and if not possible, results of abdominal ultrasound, performing a high dose dexamethasone suppression test (HDDST) or measuring endogenous ACTH may help differentiate the cause of HAC
How do I manage it?
- The cutaneous changes associated with hypercortisolemia will persist or progress unless the hypercortisolemia can be corrected. Iatrogenic hypercortisolemia should be managed by reducing the use of exogenous corticosteroids in an affected dog.
- Dogs should only be treated for HAC when there are compatible clinical signs and endocrine functions tests are diagnostic for HAC. Dogs with chronic skin disease that is clinically compatible with HAC should not be empirically treated without confirmatory endocrine function testing. Likewise, dogs with suggestive diagnostic test results but no compatible clinical signs should not be treated.
- Currently, HAC is most often managed with trilostane (Vetoryl®, Dechra Ltd) which is the only VDD approved drug for the treatment of both pituitary and adrenal dependent HAC.
- Trilostane works to reduce hypercortisolemia through competitive inhibition of the adrenal gland enzyme 3-ß-hydroxysteroid dehydrogenase thus inhibiting adrenal gland steroidogenesis and the production of cortisol.
- The dose is based on the most appropriate pill size for the dog’s body weight based on a typical starting dose range from 2.2 to 6.7 mg/kg once a day. Treatment must be individualized.
- Trilostane should not be administered to dogs with primary hepatic disease or renal insufficiency. It should never be given to pregnant animals as it is shown to have teratogenic effects in laboratory animals. The most common adverse effects of trilostane include diminished appetite, vomiting, lethargy and weakness. The most common adverse reactions reported are poor/reduced appetite, vomiting, lethargy/dullness, diarrhea and weakness. Occasionally, more serious reactions, including severe depression, hemorrhagic diarrhea, collapse, hypoadrenocortical crisis or adrenal necrosis/rupture may occur, and may result in death.
- Mitotane, o,p’DDD (Lysodren) was for many years the preferred treatment for HAC and it is still commonly used. The off label use of this drug to treat HAC is effective because of the selective adrenocortical necrosis and atrophy of the zona fasciculata (cortisol secreting layer of the adrenal cortex) and the zona reticularis (sex steroid hormone secreting layer of the adrenal cortex). The zona glomerulosa (mineralocorticoid secreting layer of the adrenal cortex) is less sensitive to the necrotizing effect of this drug but high doses can cause complete necrosis of all layers of the adrenal cortex. There are different published protocols and readers are referred to veterinary internal medicine textbooks.
- Mitotane adverse effects can develop during both induction and maintenance therapy and are most often associated with a deficiency in cortisol and patients improve when given glucocorticoid supplementation. In severe cases a hypoadrenocortical crisis with both glucocorticoid and mineralocorticoid deficiency occurring (Addisonian crisis) is possible.
- Other therapies that have been utilized in managing HAC include L-deprenyl, ketoconazole, radiation therapy of pituitary tumors, adrenalectomy or Anipryl. L-Deprenyl is VDD approved for management of mild cases of pituitary dependent HAC. It is a selective, irreversible monoamine oxidase inhibitor which results in increases in central dopamine concentrations which will have negative impact on corticotrophin release from the pars intermedia. However, fewer than 30% of dogs with HAC have a pituitary adenoma involving the pars intermedia so this treatment option has limited efficacy.
- All medical treatments involve careful monitoring of patients to determine that there is adequate control of the HAC without producing iatrogenic hypoadrenocorticism. This is done by evaluating success of treatment based on results of an ACTH stimulation test. When to perform the ACTH test and its interpretation will vary depending on the chosen medical management. There are many good internal medicine textbooks that can provide more detailed information in regards to the various treatment protocols.
- If pyoderma and/or otitis externa are present they should be managed with appropriate antimicrobial therapy based on cytology and bacterial culture and susceptibility testing.
Comments
- Lesions of calcinosis cutis typically resolve in time if the hypercortisolemia can be resolved or managed. In some cases osseous metaplasia can occur. The resulting osteoma cutis lesions will not regress.
- It is important to remember that hypercortisolemia (endogenous or exogenous) can alter thyroid function. Dogs with hypercortisolemia will often have low or below reference range serum concentrations of total thyroxine and less frequently free thyroxine is also low. Endogenous TSH in dogs with HAC is usually also low or normal. If a dog is presented with clinical signs such as alopecia, weight gain and hypercholesterolemia that could be compatible with either hypothyroidism or HAC, HAC should be screened for first before interpreting low thyroid hormone values.
- Feldman EC and Nelson RW. Hyperadrenocorticism in Canine and Feline Endocrinology and Reproduction
- Melian C., Perez-Alenza M.D, and Peterson ME. Hyperadrenocortorcism In Textbook of Veterinary Internal Medicine. 7th ed. Ed Ettinger SJ & Feldman EC: St Louis Missouri: Saunders Elsevier; 2010 p.1816-1840.
- Miller WH, Griffen DE, Campbell KL. Muller and Kirk's Small Animal Dermatology, 7th ed. St. Louis, Missouri:Elsevier Mosby 2013 p. 513-526
- Dechra: Trilostane Capsules for Dogs. For Veterinary Use Only.
Manufacturer: Dechra Ltd., Dechra House, Jamage Industrial Estate, Talke Pits, Stoke-on-Trent, ST7 1XW, UK
Distributed by: Dechra N.-A. Inc., 2000 ch. Georges, Lavaltrie, QC, J5T 3S5, Canada
VETORYL is a registered trademark of Dechra, Ltd.
abscess
A discrete swelling containing purulent material, typically in the subcutis
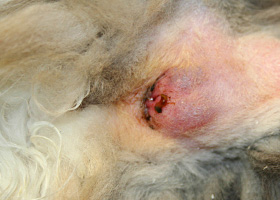
Perianal abscess in a dog
alopecia
Absence of hair from areas where it is normally present; may be due to folliculitis, abnormal follicle cycling, or self-trauma
Extensive alopecia secondary to cutaneous epitheliotropic lymphoma
alopecia (“moth-eaten”)
well-circumscribed, circular, patchy to coalescing alopecia, often associated with folliculitis
“Moth-eaten” alopecia secondary to superficial bacterial folliculitis
hemorrhagic bullae
Blood-filled elevation of epidermis, >1cm
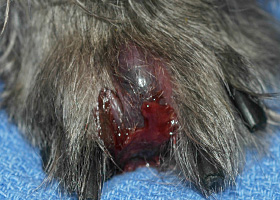
Interdigital hemorrhagic bulla in a dog with deep pyoderma and furunculosis
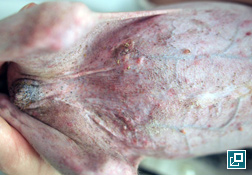
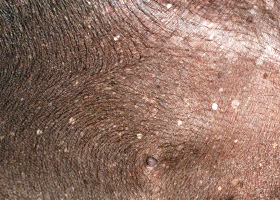
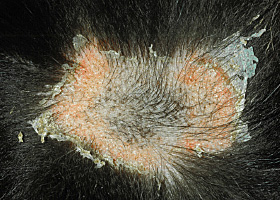
comedo
dilated hair follicle filled with keratin, sebum
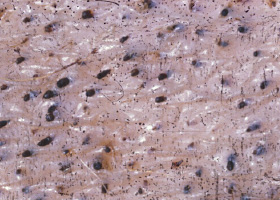
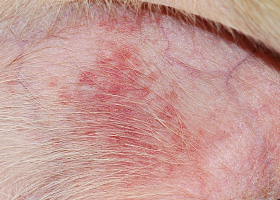
Comedones on the ventral abdomen of a dog with hypercortisolism
crust
Dried exudate and keratinous debris on skin surface
Multifocal crusts due to pemphigus foliaceus
epidermal collarettes
Circular scale or crust with erythema, associated with folliculitis or ruptured pustules or vesicles
Epidermal collarettes in a dog with Staphylococcus superficial bacterial folliculitis
erosion
Defect in epidermis that does not penetrate basement membrane. Histopathology may be needed to differentiate from ulcer.
Erosions in a dog with vasculitis
erythema
Red appearance of skin due to inflammation, capillary congestion
Erythema in a dog with cutaneous drug eruption
eschar
Thick crust often related to necrosis, trauma, or thermal/chemical burn
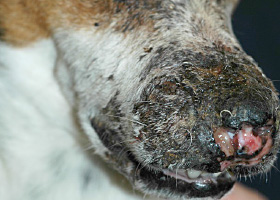
Eschar from physical trauma
excoriation
Erosions and/or ulcerations due to self-trauma
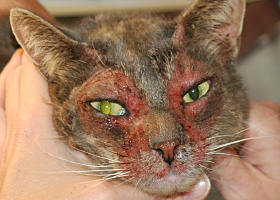
Excoriations in a cat with atopic dermatitis
fissure
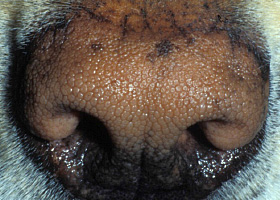
Excessive stratum corneum, confirmed via histopathology. This term is often used to describe the nasal planum and footpads.
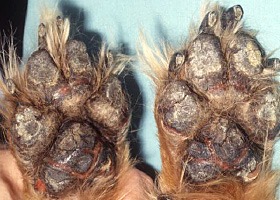
Fissures of the footpads in a dog with superficial necrolytic dermatitis
fistula
Ulcer on skin surface that originates from and is contiguous with tracts extending into deeper, typically subcutaneous tissues
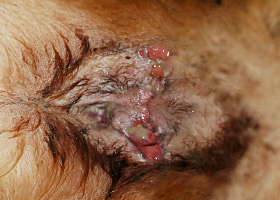
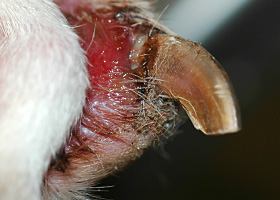
Perianal fistulas in a dog
follicular casts
Accumulation of scale adherent to hair shaft
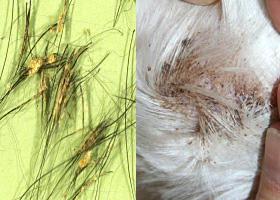
Follicular casts surrounding hairs from a dog with hypothyroidism
hyperkeratosis
Excessive stratum corneum, confirmed via histopathology. This term is often used to describe the nasal planum and footpads.
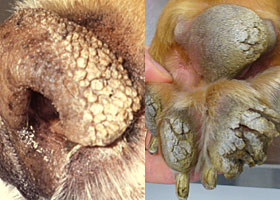
Idiopathic hyperkeratosis of the nasal planum (left) and footpads (right)
hyperpigmentation
Increased melanin in skin, often secondary to inflammation
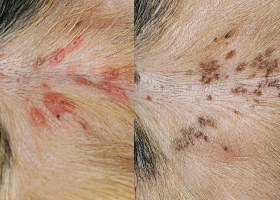
Inflammatory lesions (left) resulting in post-inflammatory hyperpigmentation (right)
hypotrichosis
Lack of hair due to genetic factors or defects in embryogenesis.

Congenital hypotrichosis in chocolate Labrador puppies.
lichenification
Thickening of the epidermis, often due to chronic inflammation resulting in exaggerated texture
Lichenification of skin in a dog with chronic atopic dermatitis and Malassezia dermatitis
macule
Flat lesion associated with color change <1cm
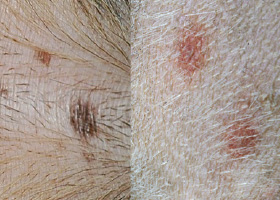
Pigmented macule (left) Erythematous macule (right)
melanosis
Increased melanin in skin, may be secondary to inflammation.
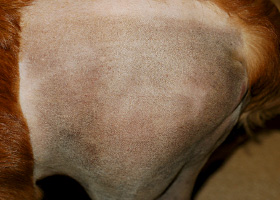
Post inflammatory hyperpigmentation of this dog’s thigh
miliary
Multifocal, papular, crusting dermatitis; a descriptive term, not a diagnosis
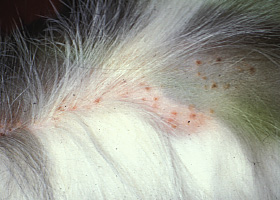
Miliary dermatitis in a flea allergic cat
morbiliform
A erythematous, macular, papular rash; the erythematous macules are typically 2-10 mm in diameter with coalescence to form larger lesions in some areas
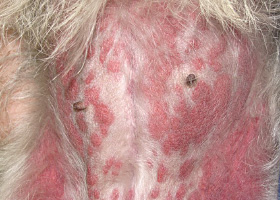
Morbiliform eruptions in a dog with a cutaneous drug reaction
onychodystrophy
Abnormal nail morphology due to nail bed infection, inflammation, or trauma; may include: Onychogryphosis, Onychomadesis, Onychorrhexis, Onychoschizia
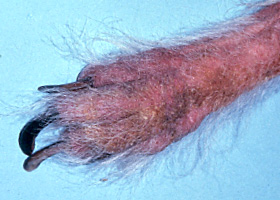
Onychodystrophy in dog with chronic allergies
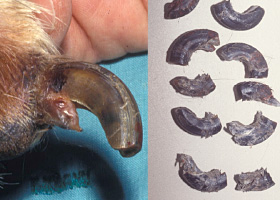
onychogryphosis
Abnormal claw curvature; secondary to nail bed inflammation or trauma

Onychogryphosis in a dog with symmetric lupoid onychodystrophy
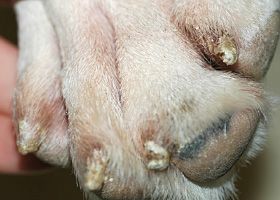
onychomadesis
Claw sloughing due to nail bed inflammation or trauma
Onychomadesis in a dog with symmetric lupoid onychodystrophy
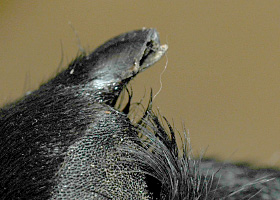
onychorrhexis
Claw fragmentation due to nail bed inflammation or trauma
Onychorrhexis in a dog with symmetric lupoid onychodystrophy
onychoschizia
Claw splitting due to nail bed inflammation or trauma
Onychoschizia in a dog with symmetric lupoid onychodystrophy
patch
Flat lesion associated with color change >1cm
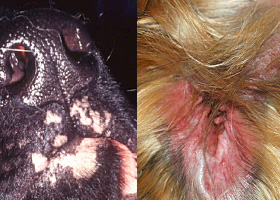
Hypopigmented patch (left), erythematous patch (right)
petechiae
Small erythematous or violaceous lesions due to dermal bleeding
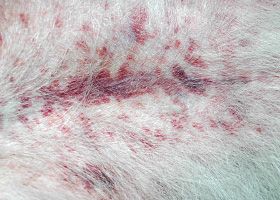
Petechiae in a dog with cutaneous vasculitis
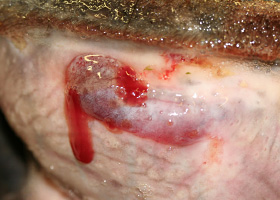
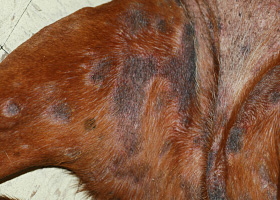
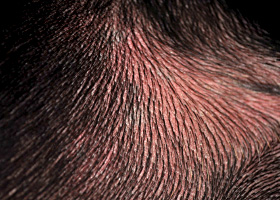
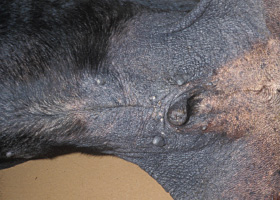
phlebectasia
Venous dilation; most commonly associated with hypercortisolism
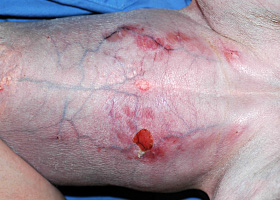
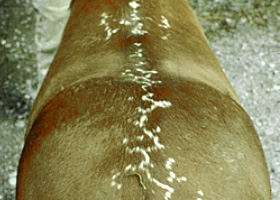
Phlebectasia and cutaneous atrophy due to hypercortisolism in a dog
plaques
Flat-topped elevation >1cm formed of coalescing papules or dermal infiltration
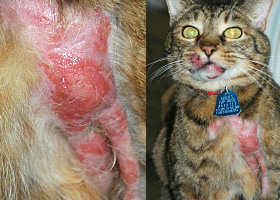
Plaques in a cat with cutaneous lymphoma
pustule
Raised epidermal infiltration of pus
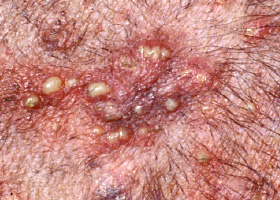
Pustules on the abdomen of a dog with superficial staphylococcal pyoderma.
scale
Accumulation of loose fragments of stratum corneum
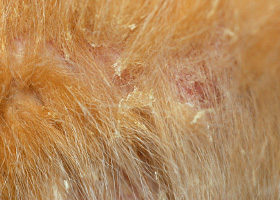
Loose, large scales due to ichthyosis in a Golden Retriever
scar
Fibrous tissue replacing damaged cutaneous and/or subcutaneous tissues
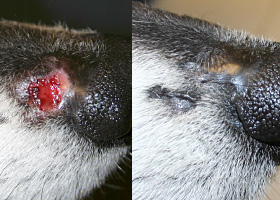
Scarring (right) following the healing of an ulcer (left) in a dog with sterile nodular dermatitis
serpiginous
Undulating, serpentine (snake-like) arrangement of lesions

Serpiginous urticarial lesions on a horse
telangiectasia
Permanent enlargement of vessels resulting in a red or violet lesion (rare)
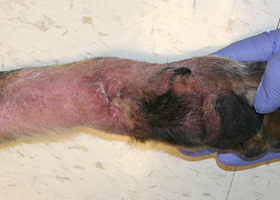
Telangiectasia in a dog with angiomatosis
ulcer
A defect in epidermis that penetrates the basement membrane. Histopathology may be needed to differentiate from an erosion.
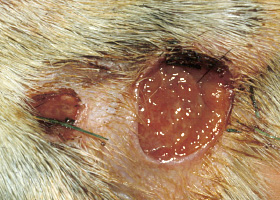
Ulcerations of the skin of a dog with vasculitis.
urticaria
Wheals (steep-walled, circumscribed elevation in the skin due to edema ) due to hypersensitivity reaction

Urticaria in a horse
vesicle
Fluid-filled elevation of epidermis, <1cm
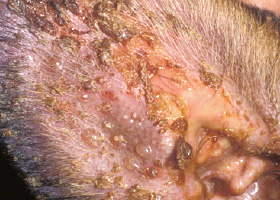
Vesicles and bullae on ear pinna due to bullous pemphigoid
wheal
Steep-walled, circumscribed elevation in the skin due to edema

Wheals associated with intradermal allergy testing in a horse